 Hence the answer to this question is that the coordination number of cesium in cesium chloride is $8$. Debye-Waller factors for these compounds are calculated over the temperature. Also as the ratio of $CsCl$ is also as that of sodium hence we find that the coordination number remains as $8$ in this case. The lattice dynamics of five compounds with the caesium chloride structure have been investigated using shell models. CsCl is more stable because its coordination number is 8: 8, compared to NaCl which has. 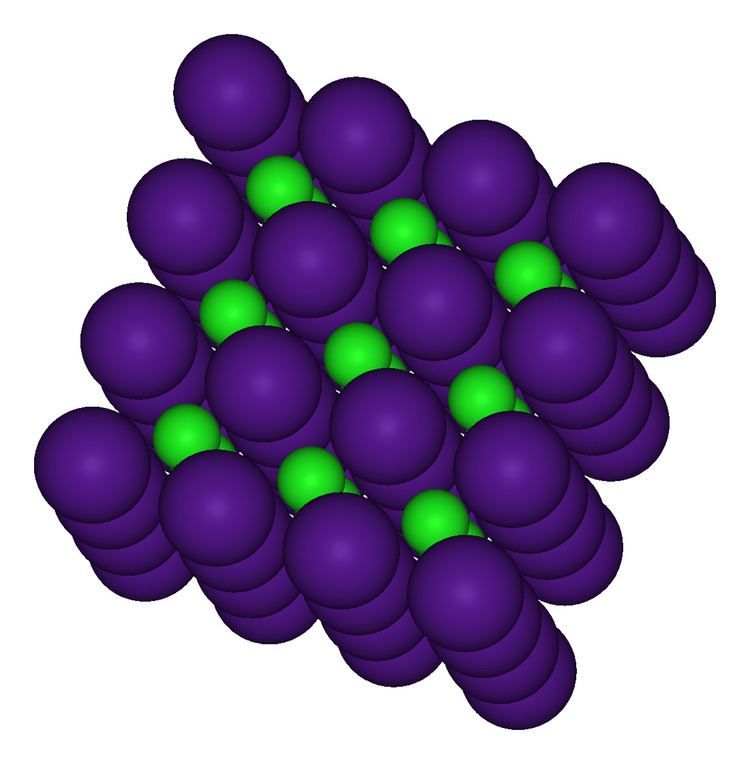
This is due to the fact that the larger the coordination number, the stronger the forces of attraction between the cations and anions in the close-packed arrangement. Hence there is a change in structure: the cesium ions do not form a lattice similar to that of sodium but rather we find that it occupies the centre of the cube that there are $8$ chloride ions surrounding it. Compare the stability of NaCl and CsCl structure. Thus we find that the size of the ion increases and hence caesium has a very large size compared to that of sodium. In case of the periodic table we are well aware that as we move down the group then the size of the cation increases and as a result of the increase in the number of the shell as we move down. 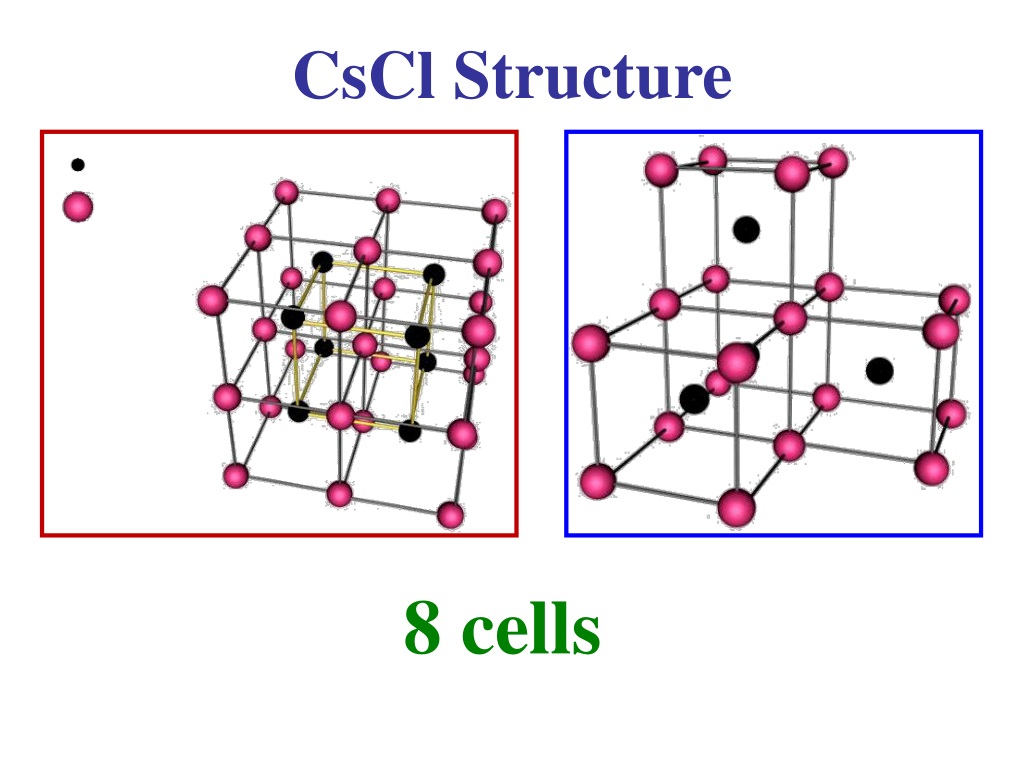
In the case of $NaCl$ the ratio is $1:1$ and as a result there are $6$ sodium ions surrounding the chloride ion and vice versa. In the case of sodium chloride we have sodium ions placed and there are $6$ chloride ions surrounding each of the sodium ions. The coordination number of a molecule is referred to as the number of ions of the other charge surrounding the ion of the opposite charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
